
Vouchers
Taking the Photos
Mollusc sampling:
Molluscs are preserved completely in pure ethanol. Depending on the amount of liquid used, you will see a more or less strong alteration of the ethanol’s color. Usually, we change the ethanol one to three times to guarantee a good fixation for high DNA yield later, a good indication is the color of the ethanol, there is no need to change it when its clear.


Mayfly sampling:
Use >95% ethanol and stainless steel, plastic, or Teflon-coated forceps to pick up the nymphs and put them in a tube (we recommend 15 ml tubes, with 1 part insects: 2 parts ethanol).
Put an individual label into each tube and write down the collecting date and name of the collector(s).
The samples are best preserved by inserting a tight-fitting piece of cotton at the top of the tube near the lid, forcing out any air bubbles, and vertical storage.
Obtain geographic coordinates (and if possible altitudes), using a portable GPS in the field and/or write down an exact description of the site to locate the position afterwards (e.g. google maps).
Please exchange the ethanol at least once a few hours after collecting (e.g. in the evening). If it is yellow or green in color we recommend changing again until the ethanol appears completely white.
Fish sampling:
Due to their size fishes require a bit more attention when sampling. Small fishes up to few centimeters can be preserved directly in either 15ml or 50ml tubes with pure ethanol, whereas larger fishes have to be sampled individually.
For our FREDIE goals, seven individually labeld specimens per species and location are ideal. Our usual procedure is as follows:
- Anesthetize and kill fishes with overdose of narcotic (MS222, Clove oil or Chlorobutanol).
- Cut the pectoral fin or part of it from the right side of the fish and put it in pure ethanol.
- Label fish with same number as tube number (we use paper stripes with the number appearing 4-times; cut & fold, then place the stripe behind the gill cover. This usually works. You can cut one of the small numbers and place it in the tube with the fin, so you don´t have to write a new label). Always put the number inside the tube, labelling on the outside will not hold for long!
- Preserve fish in a ca. 4 % formaline solution with the body as plain as possible and fins stretched out. With fomaline soaked paper towels or white fabric remnants are ideal to help to get a plain body with nicely positioned fins.
- Later, collect all fishes from one location in e.g. one plastic bag and place it in a bigger container, but please add a paper with information on date & location.
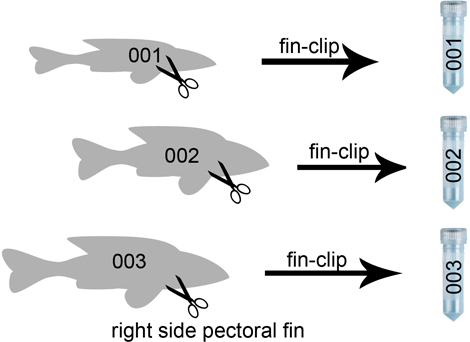

In ichthyology, dissections are always made on the right side to keep the left side intact for identification, photography, etc.
Be aware, if you work unclean and the tissue for DNA extraction comes in contact with formalin, all efforts are wasted and the tissue is lost.














































